添加微信mede1917
或扫描二维码

添加微信mede1917
或扫描二维码

欢迎来到 Medebound 国际医疗平台
平安/泰康签约的优选服务机构
欢迎来到 Medebound 国际医疗平台
平安/泰康签约的优选服务机构


 美联医邦
美联医邦
文章类型: 公司合作互动

美通社1月23日电,美国医疗评鉴机构卡思克鲁力(Castle Connolly Medical Ltd)重申同Medebound美联医邦的独家战略合作,终止与More Health的短暂合作关系。
以下为新闻中文翻译:
Castle Connolly Medical Ltd. 卡思克鲁力是位于纽约的一家医疗健康领域研究和信息的机构,以其品牌America's Top Doctors(美国顶级医生)而闻名,该公司宣布终止与More Health短暂的合作关系。
Castle Connolly卡思克鲁力将会继续与其在中国的独家合作伙伴Medebound LLC(中文名:美联医邦)合作,为中国患者和医生提供服务。美联医邦公司总裁兼首席执行官Sheena Liu博士表示,能与Castle Connolly合作感到非常荣幸。“ 我们和Castle Connolly卡思克鲁力在过去的几年中保持着非常友好和高效的合作,共同致力于服务中国市场,通过保险公司、雇主为患者提供美国顶级专家的第二诊疗意见服务,业务已经逐渐发展壮大。”
美联医邦已与中国最大的保险公司的其中三家在总部层面已经签署了重要合同(这些保险公司的规模与美国保险巨头譬如Metlife或UnitedHealth相似),100%覆盖美联医邦的远程第二意见的费用和其他辅助服务,保障和惠及了数十万名中国高端客户。
Castle Connolly卡思克鲁力总裁兼首席执行官John Connolly博士表示:“我们很高兴能继续深化与Medebound美联医邦的合作关系,并协助中国患者获得美国最顶尖医生的医疗意见。”
关于美联医邦

美联医邦®(www.medebound.com)成立于2015年,总部在纽约,是一家致力于连接中国患者和医生与美国医生的远程医疗及第二诊疗意见机构。公司开发的专利远程医疗平台既提供双语服务,又兼容美国HIPAA(健康保险交换与责任法案)隐私条例,确保了中国患者/医生和美国顶级医生之间进行影像、病历翻译,传输和远程咨询的安全性和高效性。通过与Castle Connolly Top Doctors卡思克鲁力顶级医生的独家战略合作/股权投资关系,建立了权威可信,横跨72个专科的52,000多名美国医生的美国顶级医生资源。凭借高度差异化的医生资源,先进的双语IT平台以及在美国和中国的强有力运营团队,美联医邦在中国已经获得了可观的市场吸引力。
关于Castle Connolly Medical Ltd.

Castle Connolly Medical Ltd.(www.castleconnolly.com)卡思克鲁力是一家医疗服务调研 、并出版发行顶级医生名录的机构。成立近三十年以来,始终致力于帮助消费者在全美及各地区寻找优质可靠的医疗服务和顶级医生资源。 Castle Connolly Top Doctors拥有横跨72个专科、超过52,000名美国医生的在线数据库,这些顶级医生名录是由同行提名,并经过了由医生领导的研究团队严格筛选产生的。这个顶级医生名录可以帮助患者通过医生名字、地点、专科、医院和疾病/病情进行搜索,寻找到最好的并适合患者病情的当地或者全国最顶级的医生。 Castle Connolly是美国50多个地区杂志,同时也是很多主要的国家级杂志和报纸以及各种网络媒体平台的的顶级医生信息来源。 2017年,该网站的访问量超过1.1亿。

刘欣娜博士与克鲁力博士(左)和卡思先生(后)在纽约办公室举行的签约仪式,共同组建美联医邦董事会
新闻原文
Castle Connolly Medical Ltd., a New York based healthcare research and information company noted for its brand America’s Top Doctors®, and More Health, Inc. a California based medical second opinion company with operations in China, have ended a brief relationship. Castle Connolly will continue to work with its exclusive partner in China, Medebound LLC, in serving Chinese patients, doctors and hospitals. Sheena Liu, M.D., Ph.D., the President and CEO of Medebound, stated that she was pleased with her company’s relationship with Castle Connolly. “We have enjoyed a very positive and productive relationship with Castle Connolly through past years. Our second medical opinion business serving insurers, employers and patients has grown substantially and together we are committed to the Chinese market.” Medebound has executed important contracts with 3 of the largest insurance companies in China with size similar to Metlife or UnitedHealth in the US, at the headquarter level reaching hundreds of thousand Chinese clients whose insurance health benefits 100% cover/reimburse Medebound’s remote second opinions and other ancillary services. “We are delighted to continue to expand our relationship with Medebound and to help assist Chinese healthcare consumers seeking the expertise of the most America’s Top Doctors”,“ said Dr. John Connolly, Castle Connolly’s President and CEO
About Medebound LLC
Founded in 2015, Medebound (www.medebound.com) is a tele-health/second opinion company headquartered in New York with the focus on connecting Chinese patients/physicians with US doctors. This is achieved by creating a proprietary bi-lingual HIPAA-compliant tele-health platform, which facilitates secure medical imaging/records translation, transferring and tele-consultations between Chinese patients/local physicians and leading American specialists; and by establishing an exclusive strategic/equity relationship with Castle Connolly Top Doctors®, an established, trusted and authoritative resource for identifying America’s leading physicians, with more than 52,000 U.S. physicians across 72 medical specialties. With this highly-differentiated physician resource and first-of-its-kind bilingual IT platform and a strong execution team operating both in the US and China, Medebound has gained significant market traction.
About Castle Connolly Medical Ltd.
Castle Connolly Medical Ltd. (www.castleconnolly.com) is a healthcare research, information, and publishing company whose mission since its inception nearly three decades ago is to help consumers find independent, trusted information about quality healthcare and Top Doctors on both the regional and national levels. Castle Connolly Top Doctors®, an online database of more than 52,000 U.S. physicians across 72 medical specialties who have been nominated by their peers and rigorously screened by its physician-led research team, is used widely by focused healthcare consumers who seek the best healthcare. This database is searchable by physician name, location, specialty, hospital, and disease/condition. Castle Connolly is the source of Top Doctors’ information for more than 50 regional magazines across the U.S., many major national magazines and newspapers, and diverse online media distribution platforms. In 2017, visitors to the website generated 110 Million+ views of Castle Connolly Top Doctors detailed profiles.
相关链接:
1. https://www.prnewswire.com/news-releases/castle-connolly-medical-ltd-and-more-health-inc-mutually-terminate-their-relationship-300586299.html
2. http://markets.businessinsider.com/news/stocks/Castle-Connolly-Medical-Ltd-and-More-Health-Inc- utually-Terminate-Their-Relationship-1001689333
3. http://www.castleconnolly.com/
本文由美联医邦Medebound原创,欢迎转发,未经美联医邦允许,不得转载,违者必究。
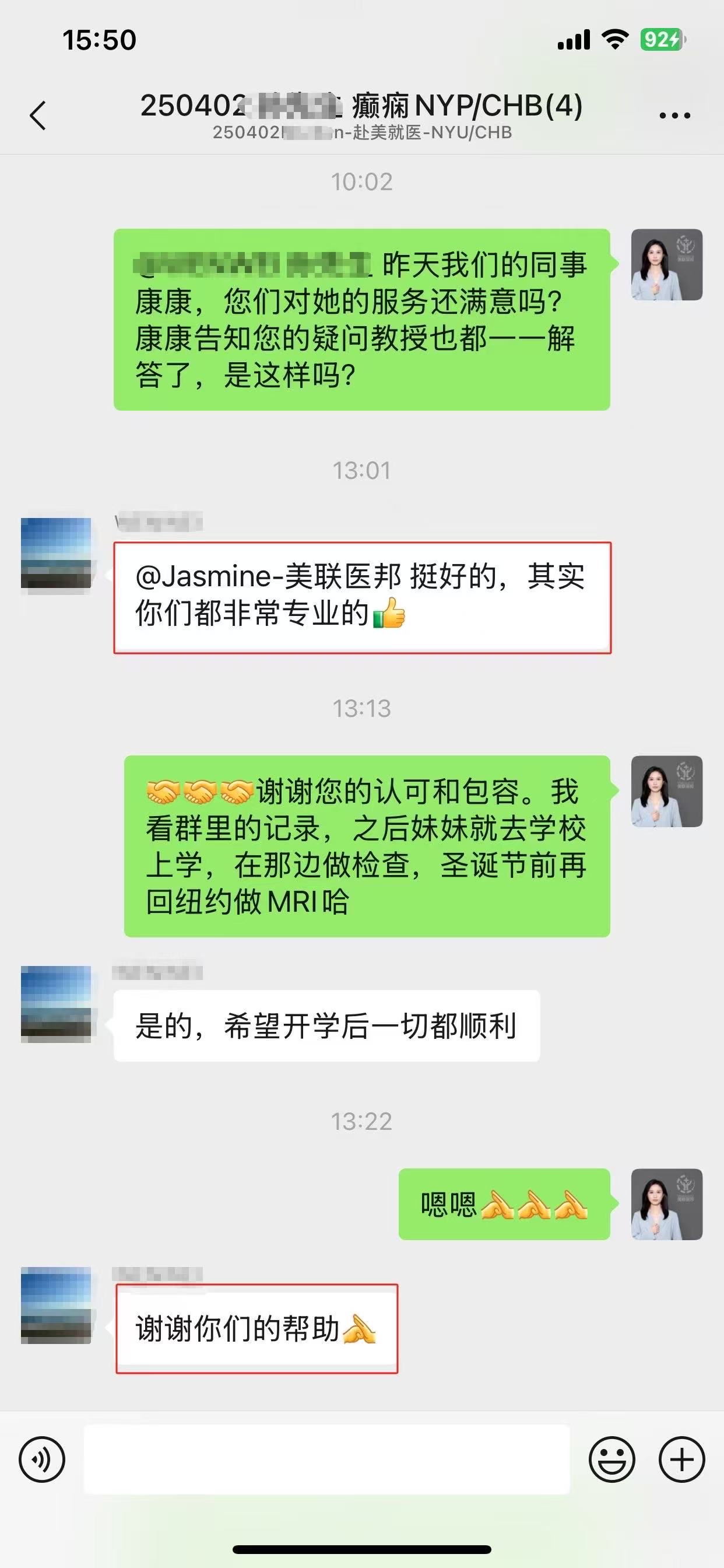
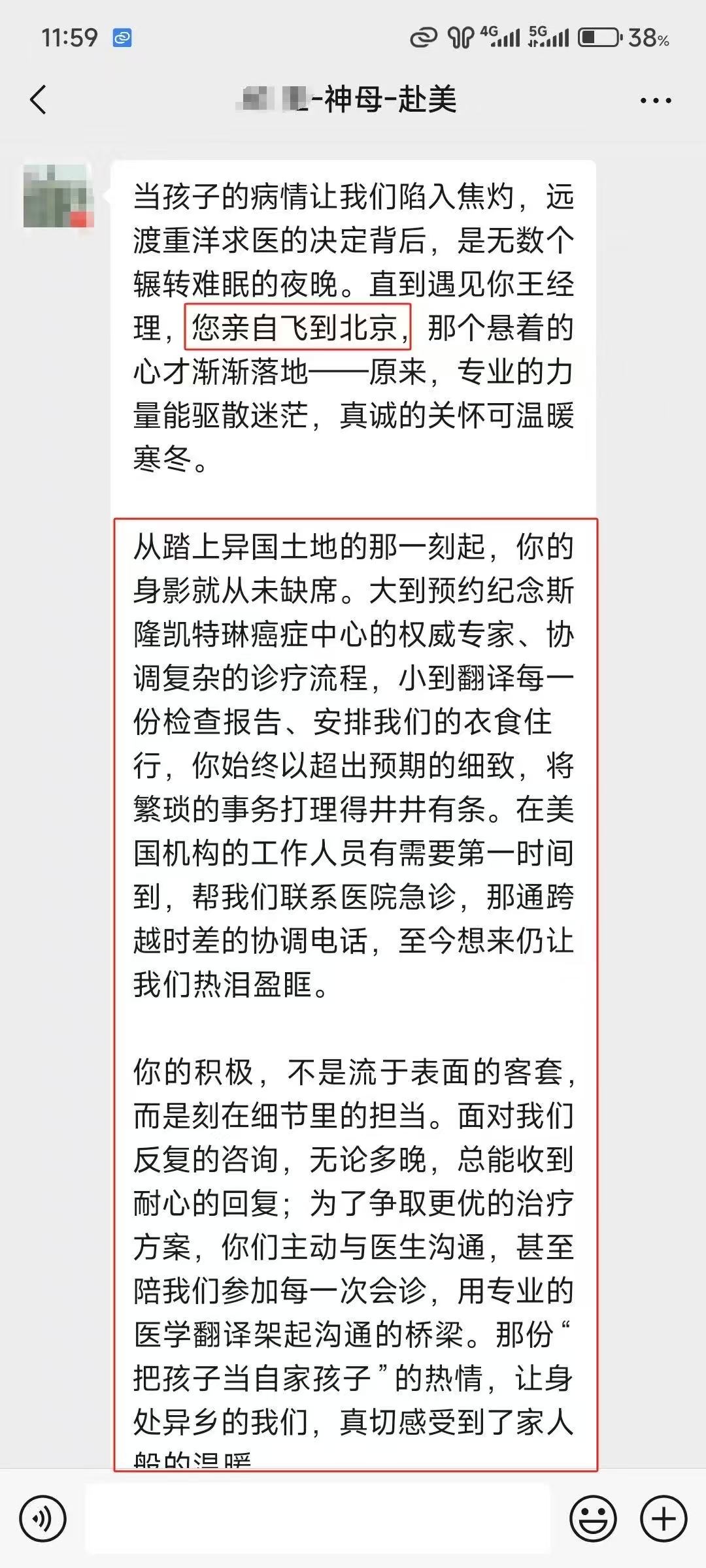
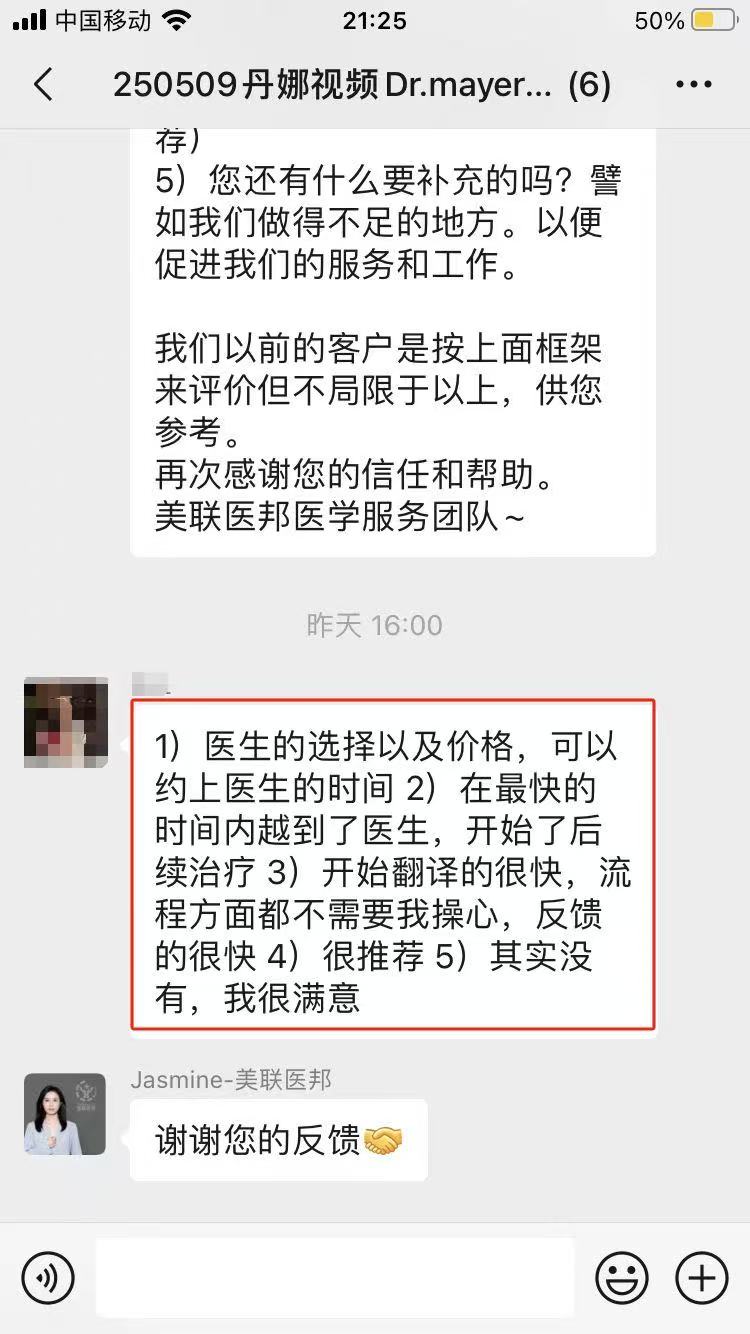
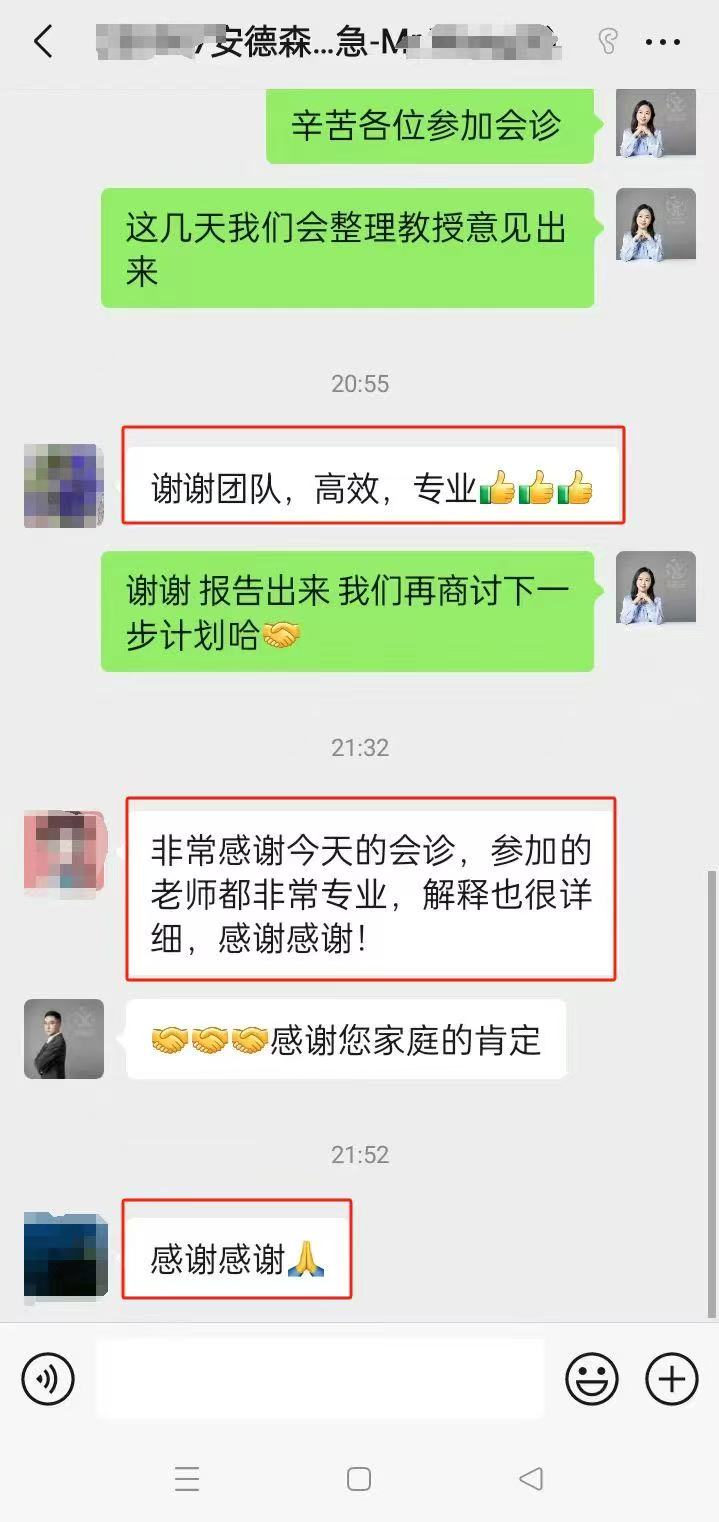
部分美联医邦美国名医真实案例:
☞中国还原体肌病患儿获美国专家免费5+小时多学科会诊、免费食宿,感恩遇见美国最高政府卫生研究院
☞膀胱癌患者通过视频会诊中美两国三地联线哈佛名医,寻求治疗方案及临床试验建议
微信公众号:medebound



凭借我们30多年的美国医疗网络资源,您可以直接与美国医疗精英对话,在家中咨询美国顶级专家或国际会诊,不出国门获取先进治疗方案。
凭借我们30年的医疗网络资源,您可以直接与美国医疗精英对话。在家中咨询疑难疾病的专家,轻松了解治疗方案。
如需赴美就医或获取海外新药,我们为您一站式安排海外看病等服务。



服务优势
预约知名美国专家譬如安德森癌症医院和梅奥诊所,安排无忧出国看病行程,寻找全球新药新技术医生网络
我们与美国顶级1%的海外医疗医生网络,300+所美国权威医院和药房资源深度合作一站式海外诊疗我们的专属客服医学经理,为您免费咨询,最短时间获取预约优质医疗资源

Jason 王经理
医学客服经理/添加微信:Mede2018

Kiki 圆经理
医学客服经理/添加微信:mede1917

如果您和您的家人有任何医疗和新药需求,请欢迎随时联系我们。我们客服团队工作时间周一到周五早上9点到晚上8点,北京时间。
总部位于纽约,9年专注美国,是赴美就医服务细分领域的头部机构。 始终坚持精英式定制(非保姆式),获30+历史卡思克鲁力医生集团(全美TOP医生联盟)及股东战略产业投资,不涉及风险资本。 作为美国本土医疗资源提供商,美联医邦已与中国平安、泰康人寿、太平人寿等保险集团达成总对总合作。服务覆盖数百万保险客户。美国福布斯榜推荐和英文报道,直通全美前5%顶级专家网络。只精准对接全美72个专科排名TOP3医院,包括梅奥诊所、MD安德森、纪念斯隆-凯特琳癌症中心等百余家美国著名医疗机构。申请美国医院的折扣率10-30%,和美国医院议价能力高。3000+客户的信任选择,一切从用户角度出发,鼓励部分患者远程二诊/问诊拿方案在国内治疗,不过度宣传和劝退不必要的赴美看病。
了解更多美国美联医邦成都运营服务中心
Disclaimer 免责声明:
本文无意影响或改变您的主治医生提供的医疗服务。请不要在没有先咨询您的主治医疗服务提供者的情况下对您的治疗做出改变。本文不用于诊断或治疗疾病,也不影响治疗方案。美联医邦会尽最大努力编辑和更新本页面的信息,但是我们无法保证本页医药信息的精确性和完整性。

纽约总部:
260 Madison Ave 8th Floor #8001,New York, NY 10016
美联医邦Medebound成都运营服务中心:
成都市锦江区红星路一段35号A区1号楼605
(美)+1 917-342-2381
(中) +86 400-616-2591
support@medebound.com